SOLVED:A solution was prepared by mixing 50.00 mL of 0.100 M HNO3 and 100.00 mL of 0.200 M HNO3 . Calculate the molarity of the final solution of nitric acid.

The student will: be able to explain the experimental technique of titration. math calculate the molarity or volume of an unknown solution using the titration. - ppt download

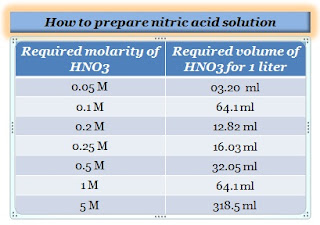
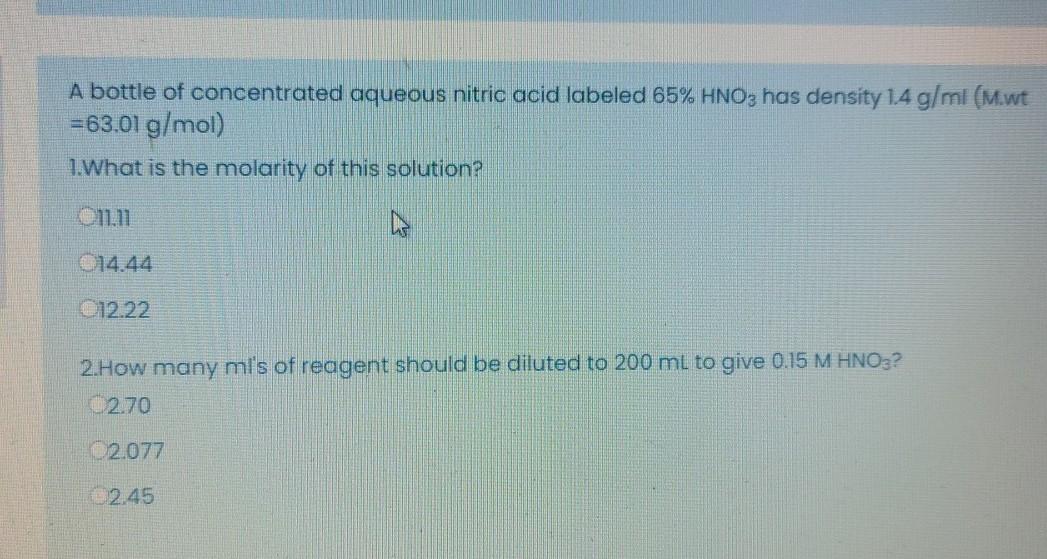
Calculate the concentration of nitric acid in moles per litre in a sample which has a density 1.41 g mL^-1 and the mass per cent of nitric acid in it being 69% .

Graph of molarity of nitric acid versus uranium extraction efficiency... | Download Scientific Diagram