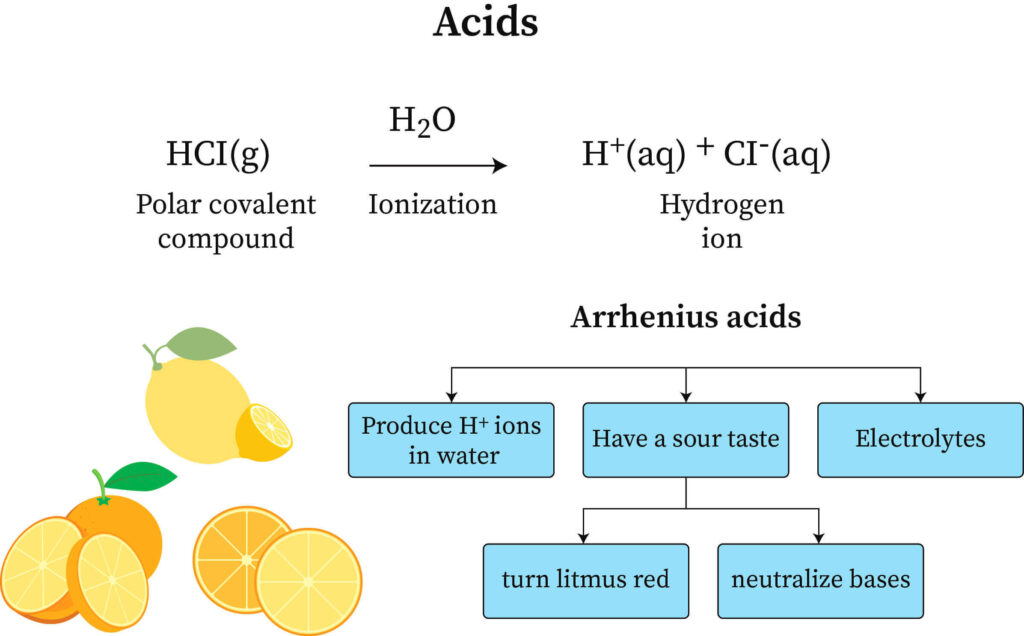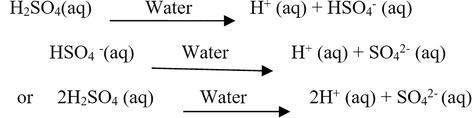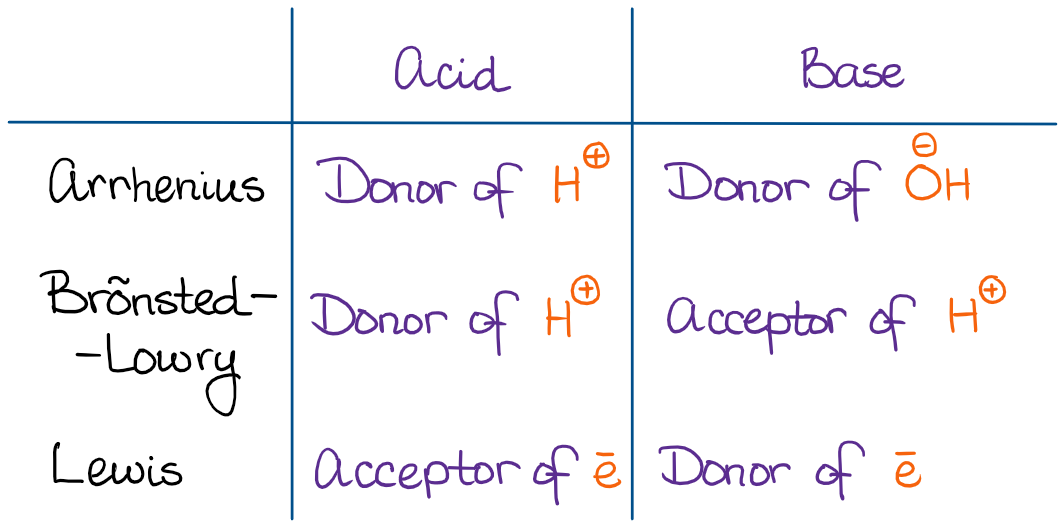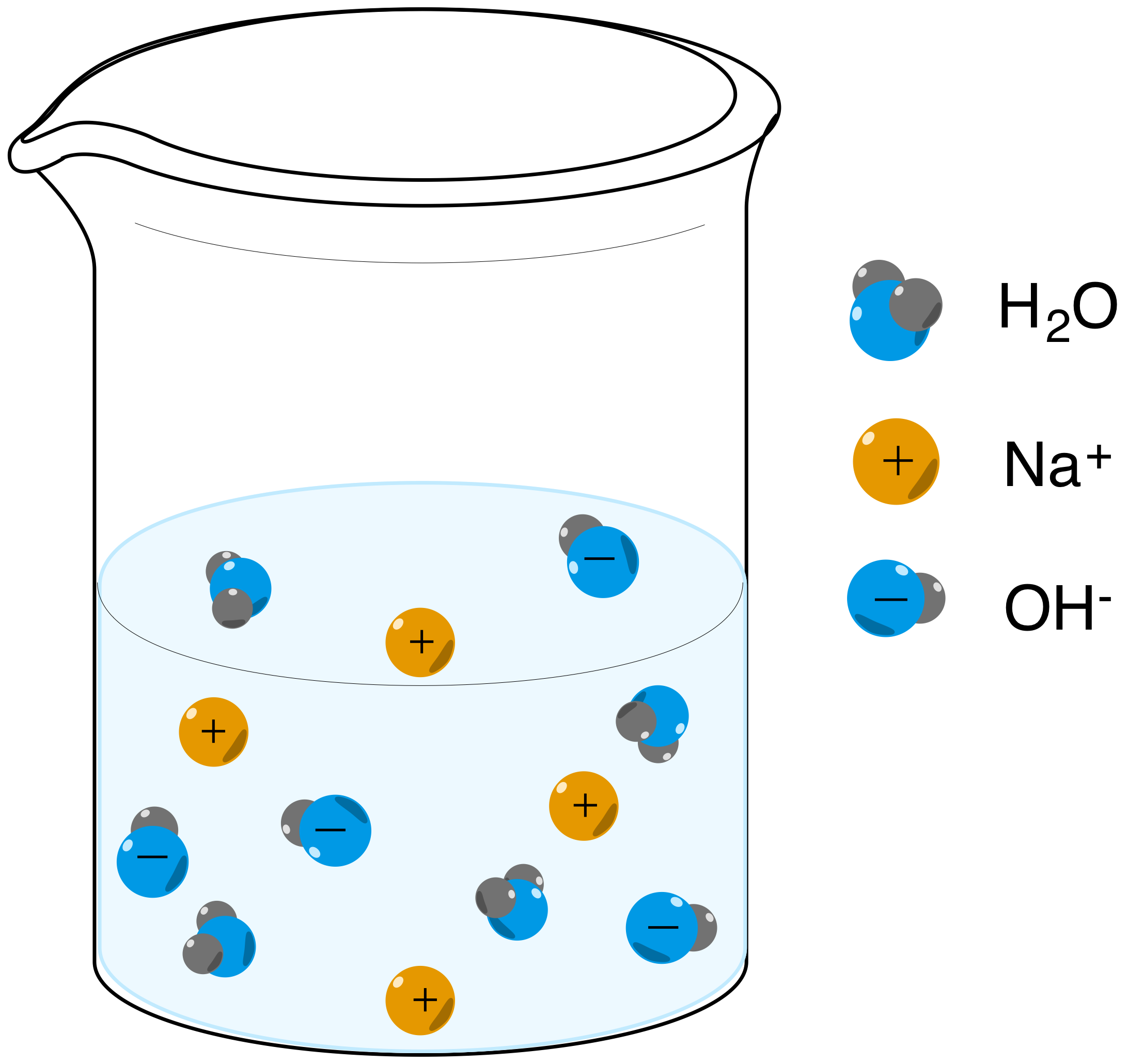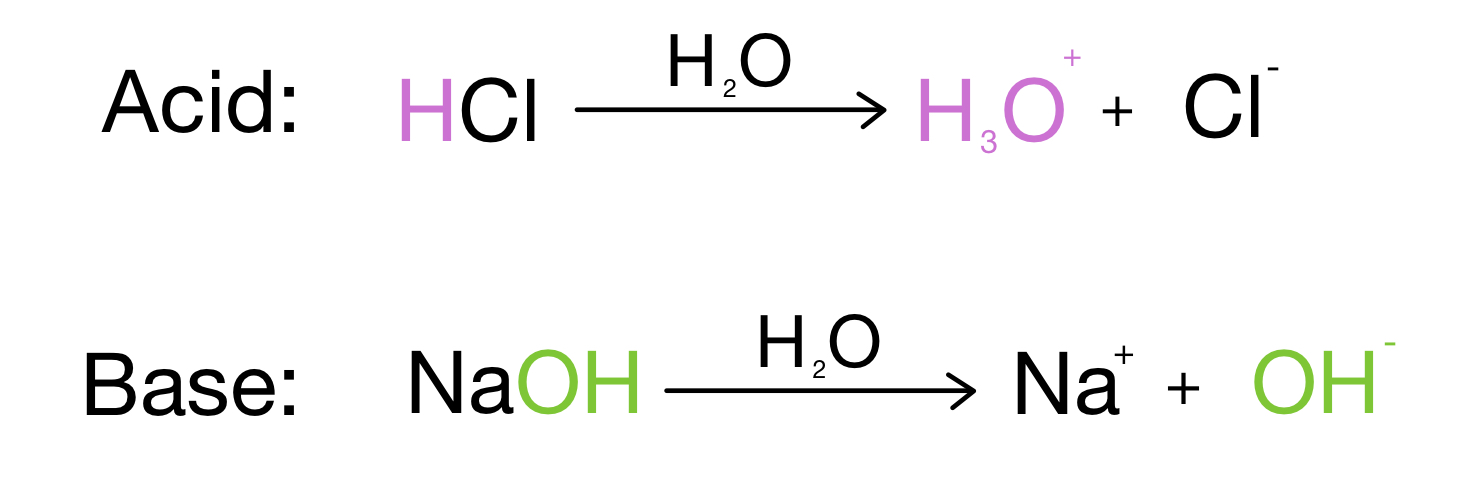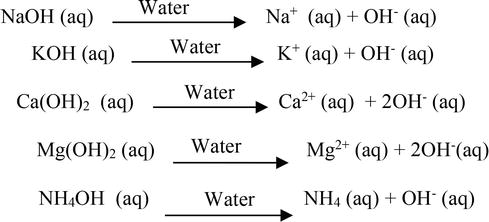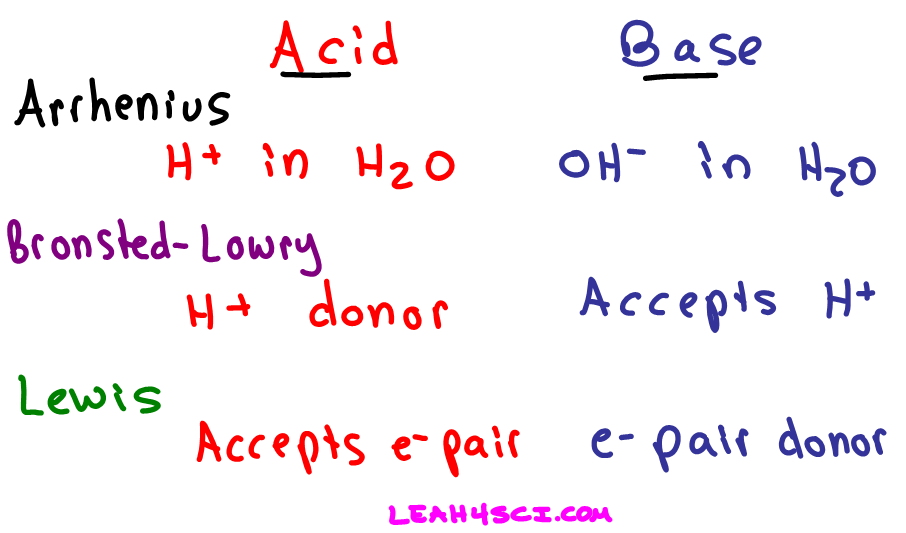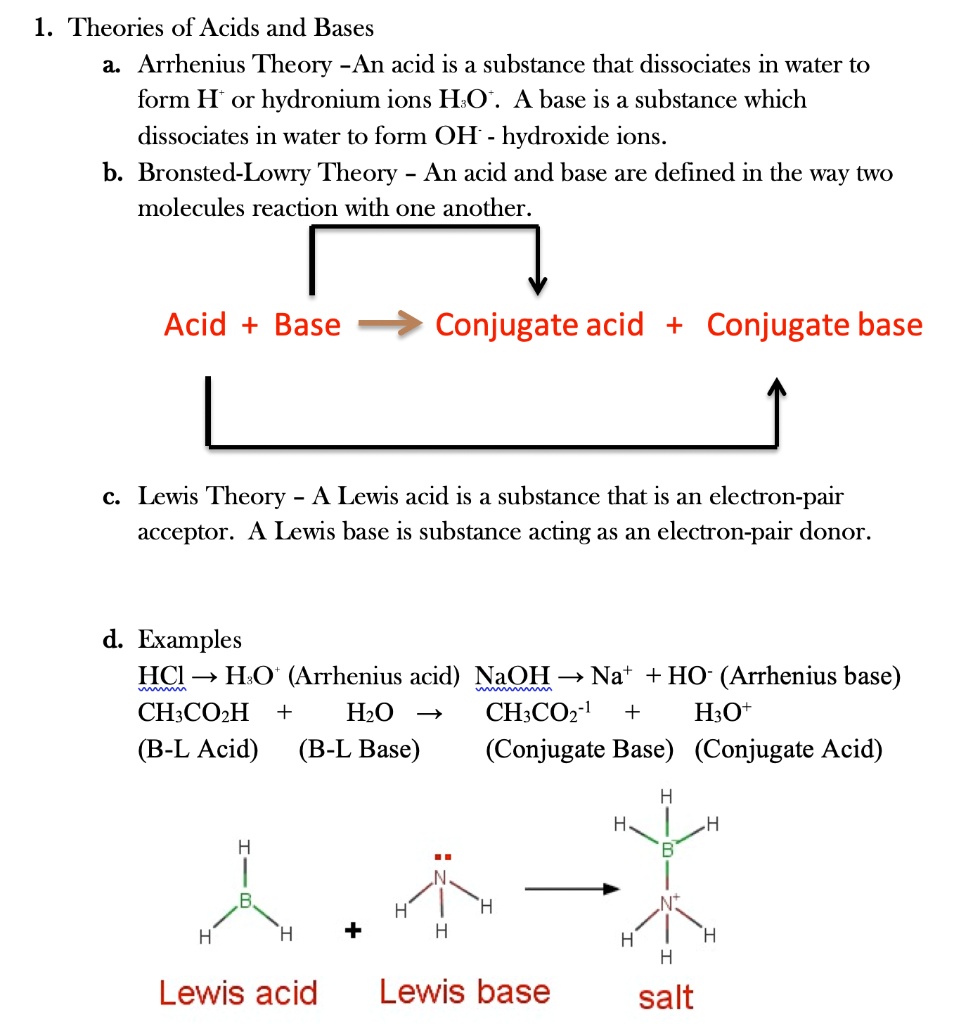
SOLVED: 1 Theories of Acids and Bases Arrhenius Theory -An acid is a substance that dissociates in water to form H or hydronium ions HO . A base is a substance which
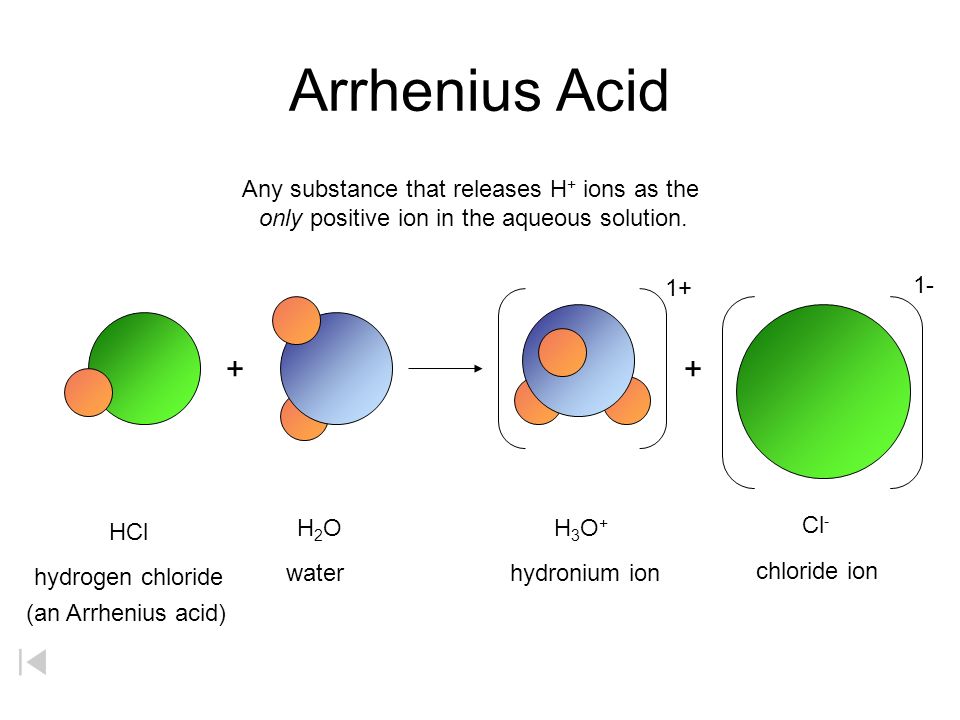
Definitions. Arrhenius Acids and Bases Acids release hydrogen ions in water. Bases release hydroxide ions in water. An acid is a substance that produces. - ppt download

.PNG)