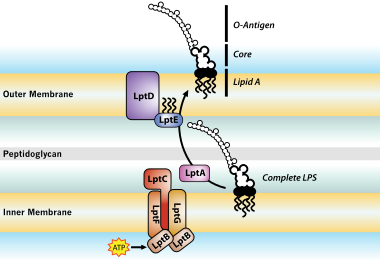
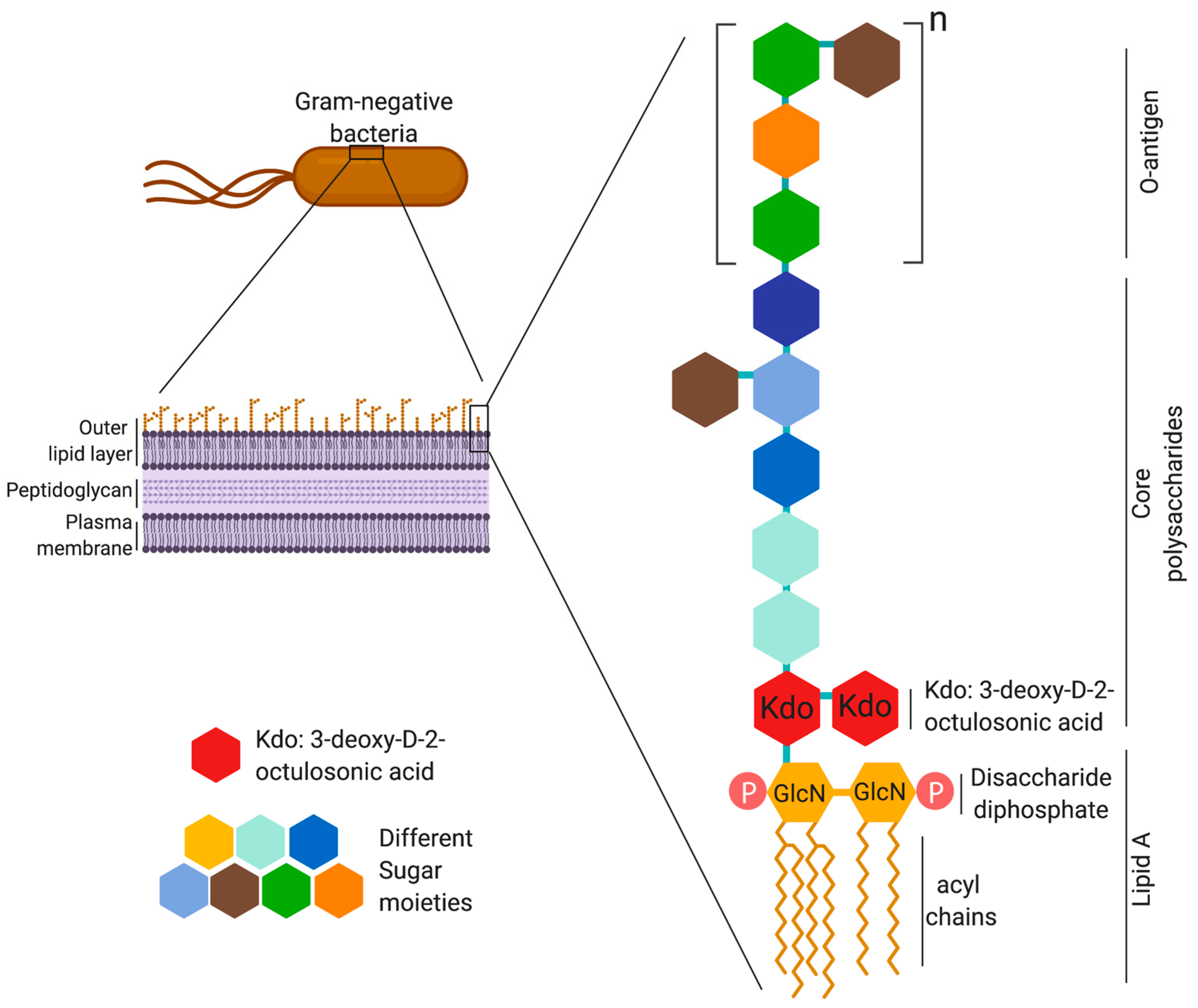
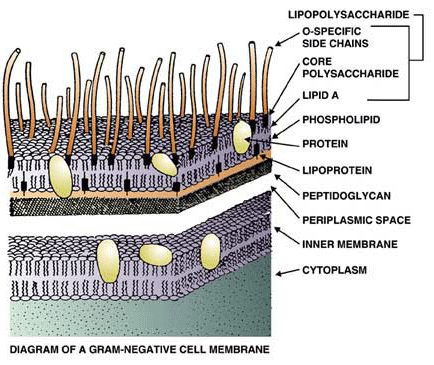
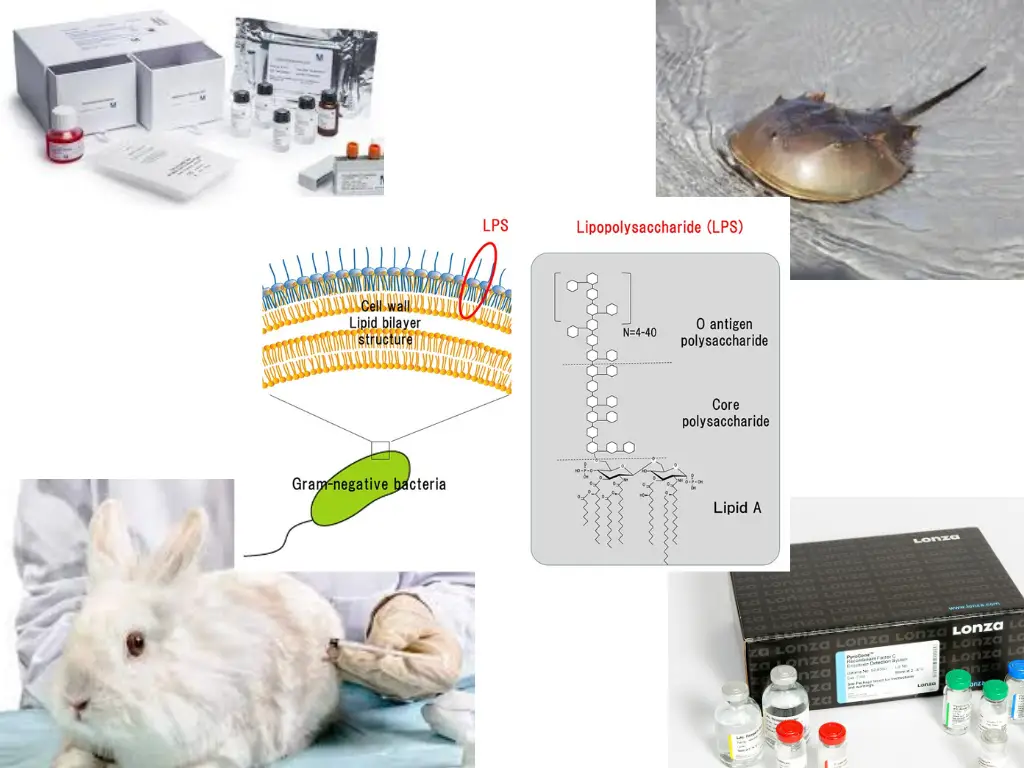
![Pyrogen Detection: Endotoxin, Peptidoglycan and β-D-Glucan | [Pharma Manufacturing & QC] | Laboratory Chemicals-FUJIFILM Wako Chemicals Europe GmbH Pyrogen Detection: Endotoxin, Peptidoglycan and β-D-Glucan | [Pharma Manufacturing & QC] | Laboratory Chemicals-FUJIFILM Wako Chemicals Europe GmbH](https://labchem-wako.fujifilm.com/europe/category/images/Pyrogen_Detection_img01.png)
Pyrogen Detection: Endotoxin, Peptidoglycan and β-D-Glucan | [Pharma Manufacturing & QC] | Laboratory Chemicals-FUJIFILM Wako Chemicals Europe GmbH
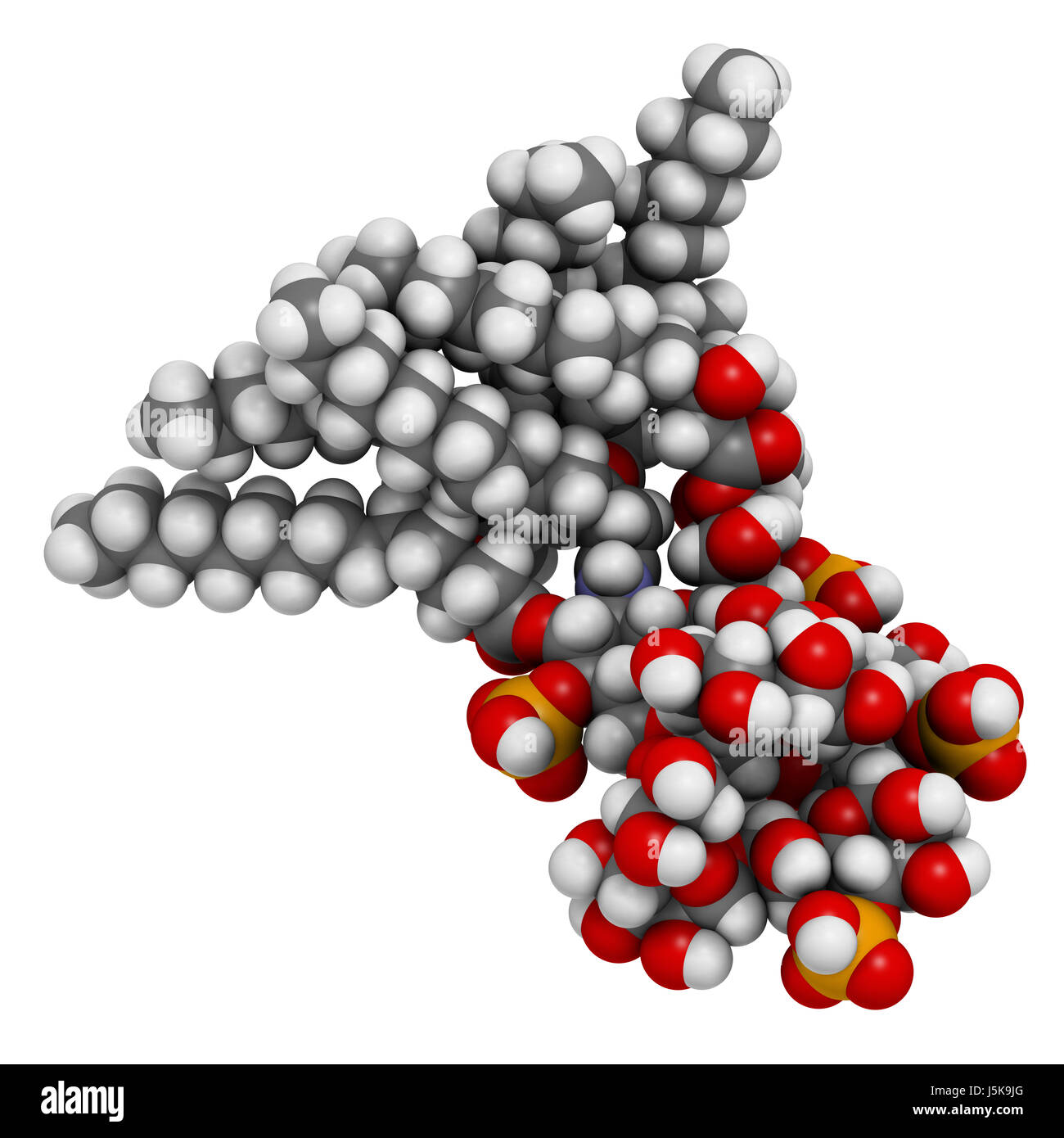
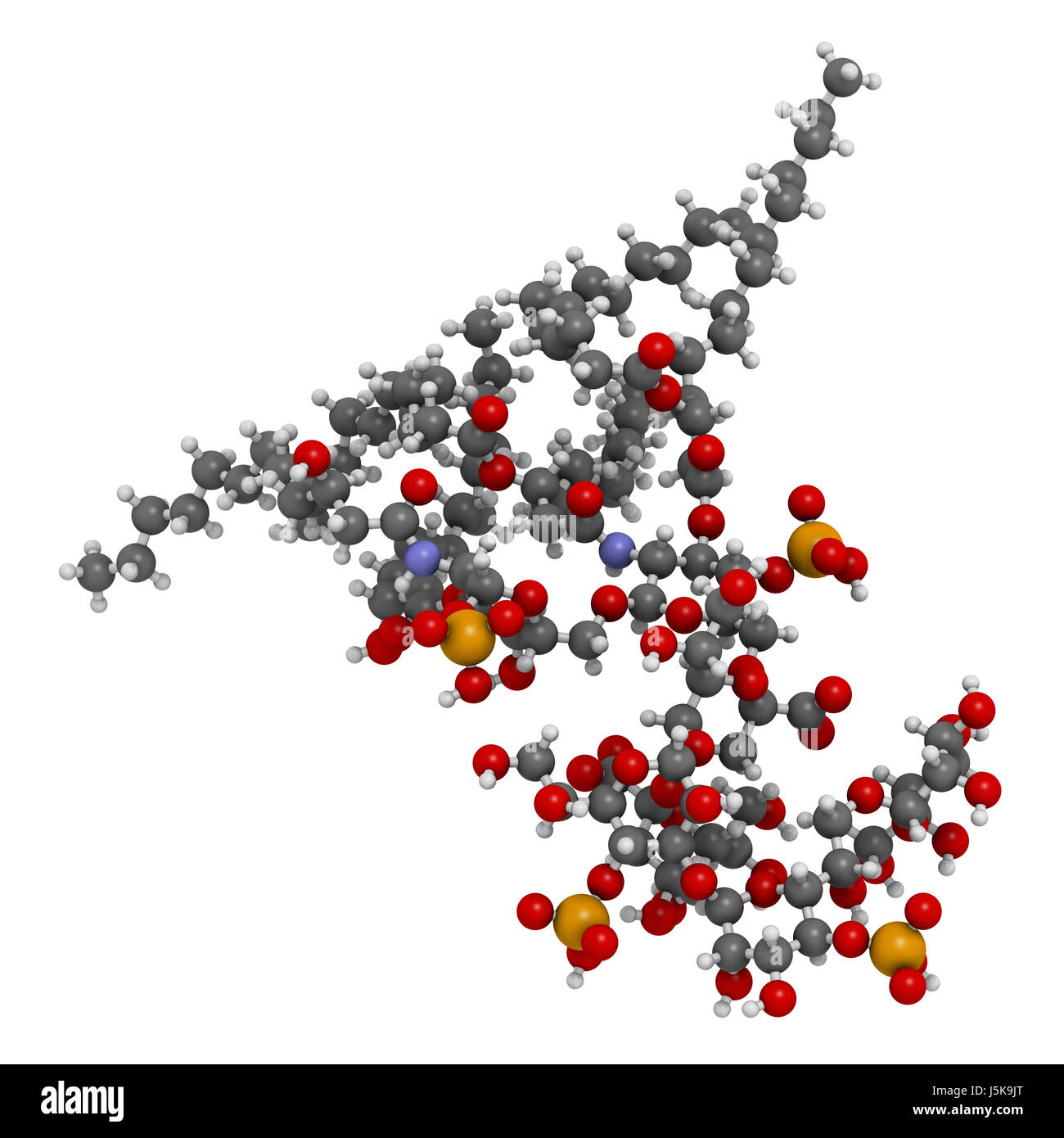
Lipopolysaccharide (LPS, lipid A and inner core fragment) endotoxin molecule from E. coli. 3D rendering based on protein data bank entry 3fxi Stock Photo - Alamy
Pyrogen Testing Revisited on Occasion of the 25th Anniversary of the Whole Blood Monocyte Activation Test. - Document - Gale OneFile: Health and Medicine
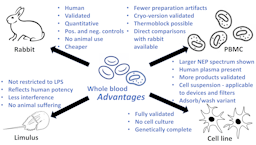
Pyrogen testing revisited on occasion of the 25th anniversary of the whole blood monocyte activation test | ALTEX - Alternatives to animal experimentation

Pyrogens, a polypeptide produces fever by metabolic changes in hypothalamus: Mechanisms and detections - ScienceDirect

Part III, Endotoxin Test Concerns of Biologics: LER From a Broad Biologics Test Perspective | American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology
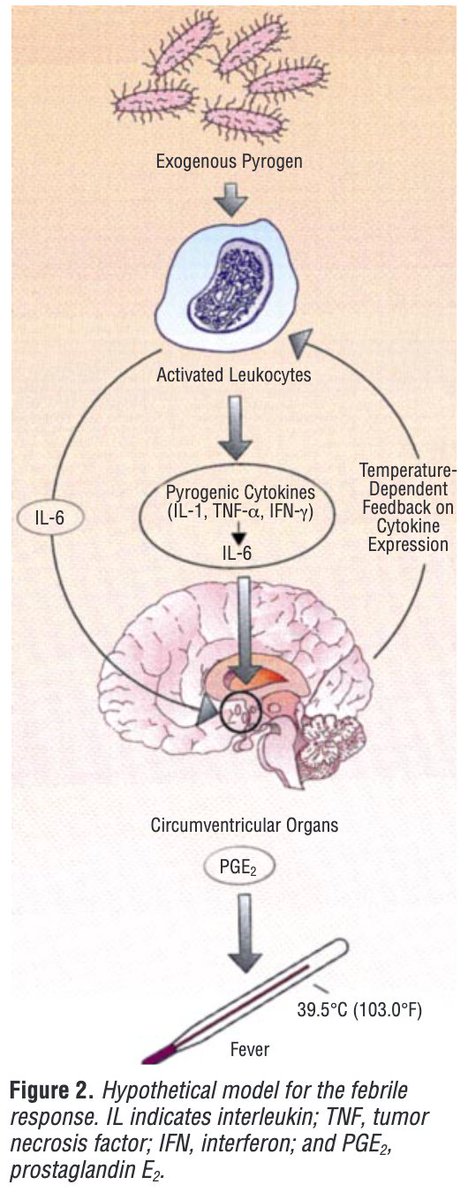
Tony Breu on Twitter: "2/ Bacteremia exposes us to exogenous pyrogens. For example, the cell wall of gram-negative rods contains lipopolysaccharide ( LPS; endotoxin). When injected into humans LPS induces fever. But, there

Pyrogens, a polypeptide produces fever by metabolic changes in hypothalamus: Mechanisms and detections - ScienceDirect