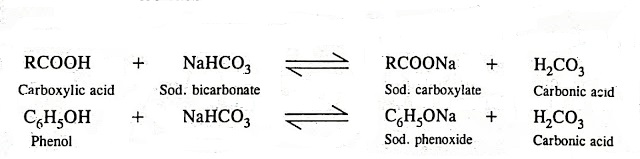
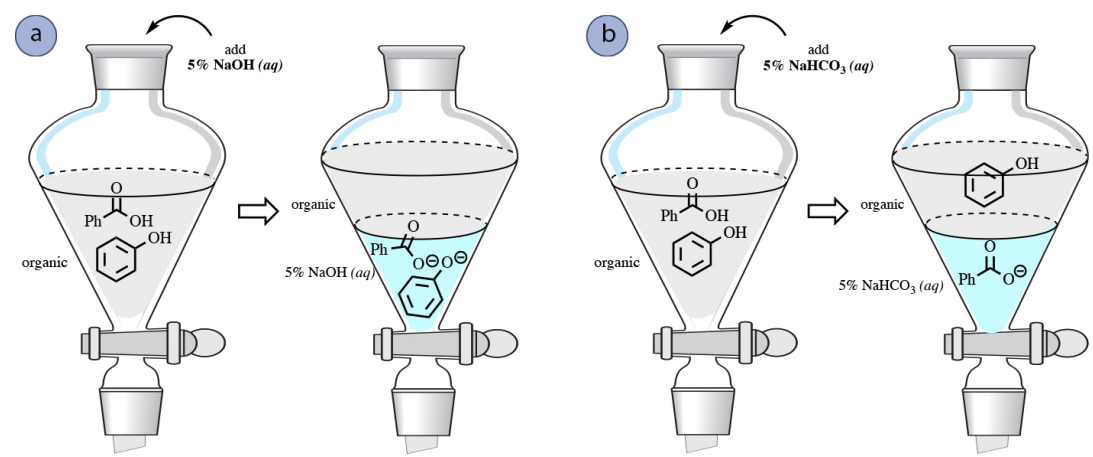
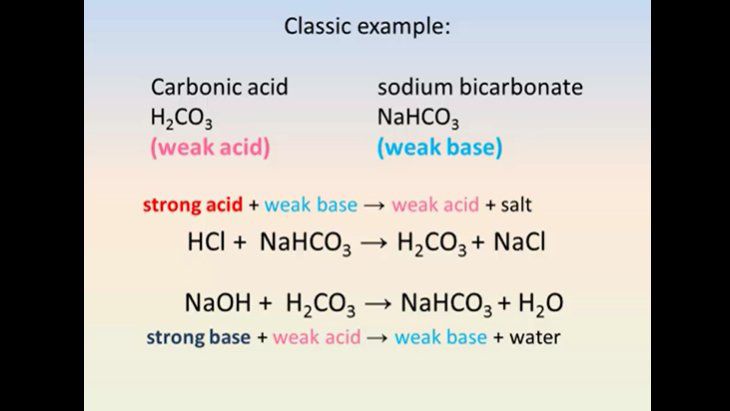
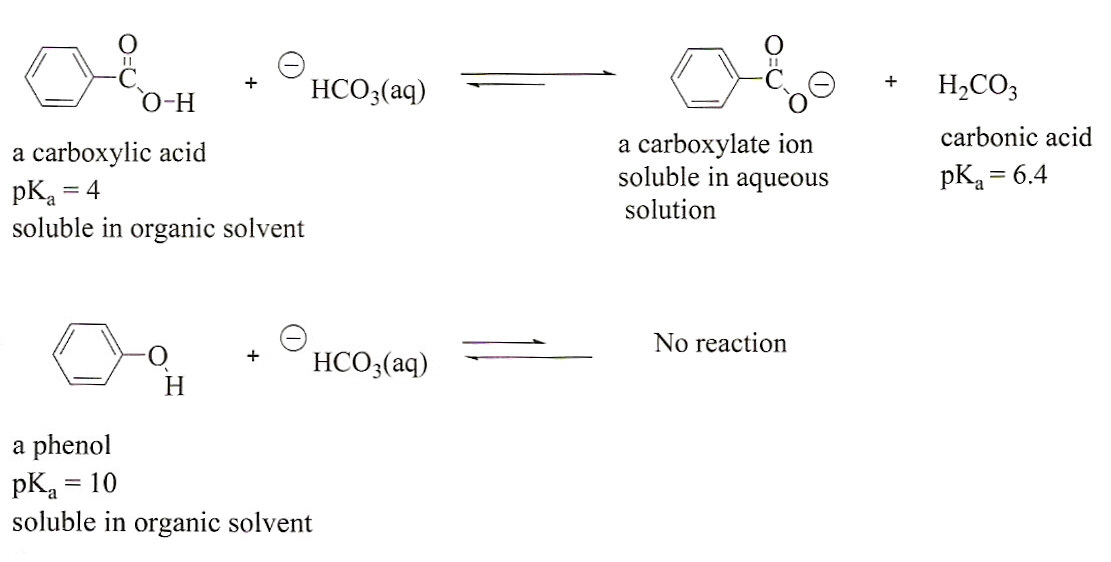
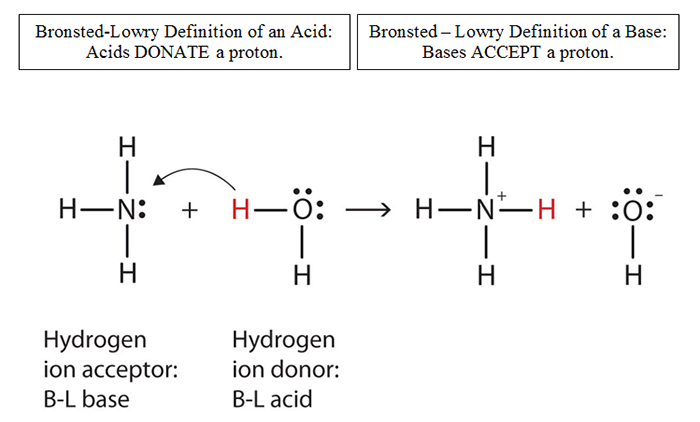
Stomach acid, a dilute solution of HCl in water, can be neutralized by reaction with sodium hydrogen carbonate. NaHCO(3((aq)))+HCl((aq)) to NaCl((eq))+H(2)O((l))+CO(2(g)) How many milliliters of 0.125 M NaHCO(3) solution are needed to
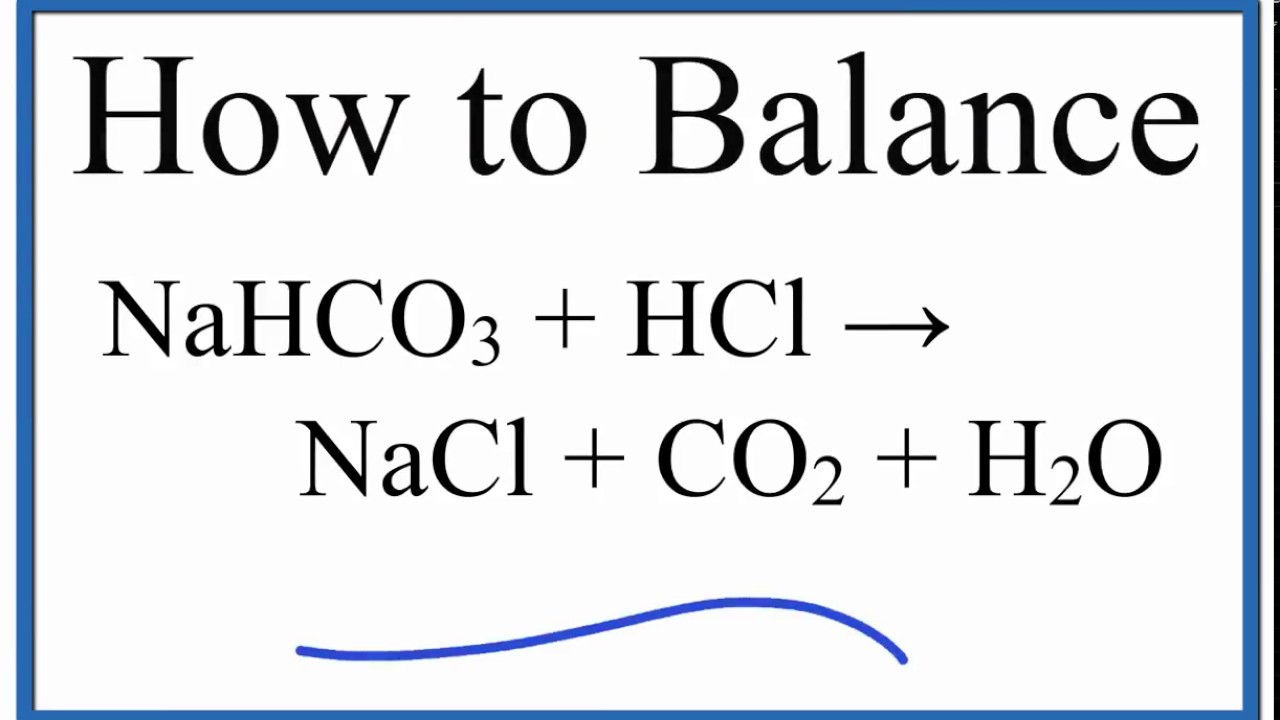
How to Balance NaHCO3 + HCl = NaCl + CO2 + H2O (sodium bicarbonate plus hydrochloric acid) - YouTube

SOLVED: How many moles and grams of acetic acid would be required if 5 grams of sodium bicarbonate reacted completely with no unreacted acid or base (i.e. with an exact stoichiometric ratio?

Using aqueous hydrochloric acid, sodium bicarbonate, or sodium hydroxide solutions, devise a flow-chart separation scheme to separate the following two-component mixtures. Both substances are soluble | Homework.Study.com