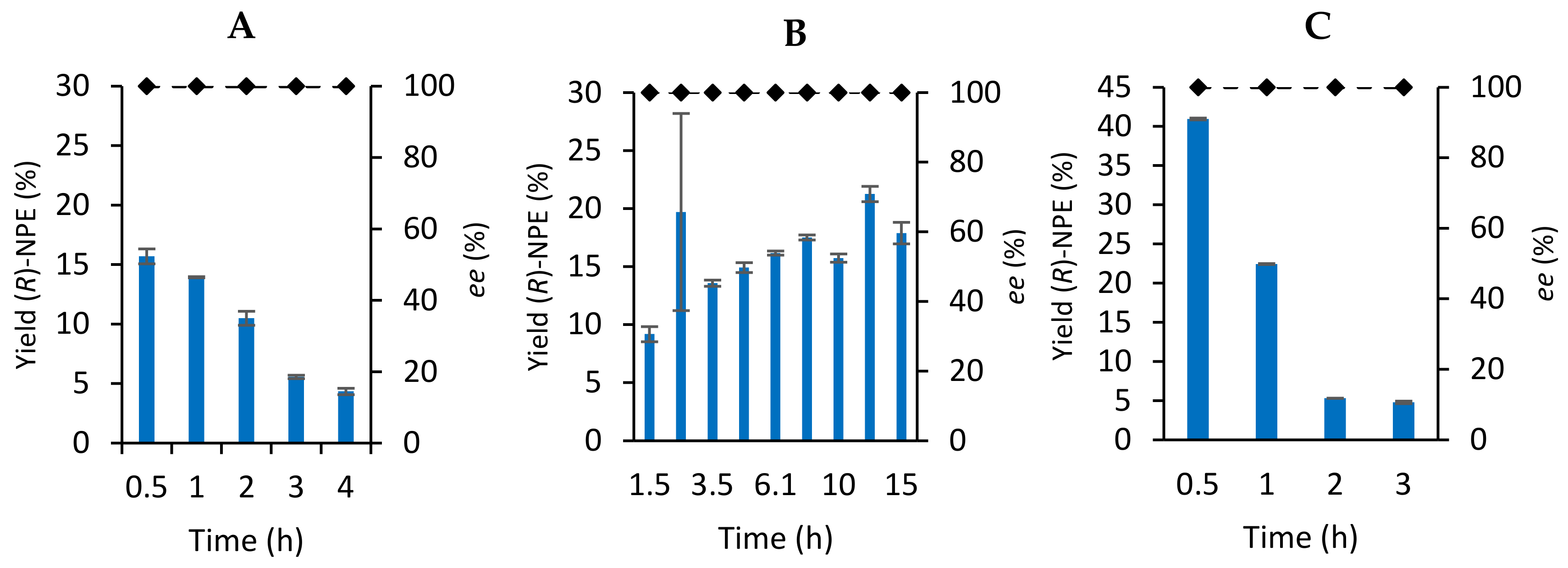
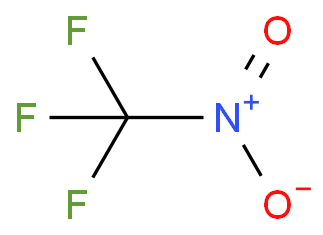
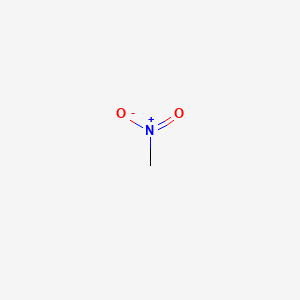
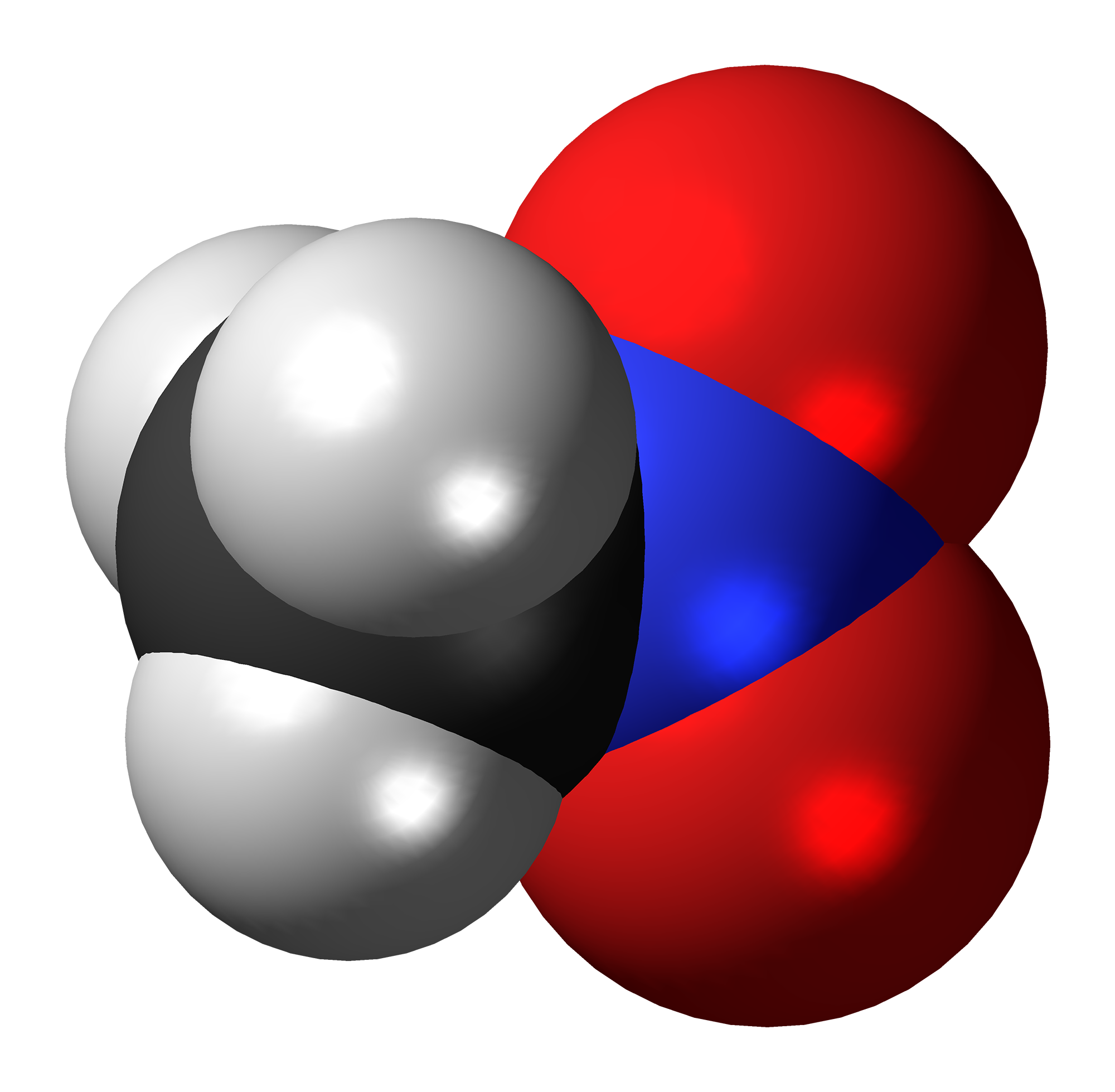
Organotitanium Nucleophiles in Asymmetric Cross-Coupling Reaction: Stereoconvergent Synthesis of Chiral α-CF3 Thioethers | Journal of the American Chemical Society
![Pharmacological Characterization of the Novel Histamine H3-Receptor Antagonist N-(3,5-Dichlorophenyl)-N′-[[4-(1H-imidazol-4-ylmethyl)phenyl]-methyl]-urea (SCH 79687) | Journal of Pharmacology and Experimental Therapeutics Pharmacological Characterization of the Novel Histamine H3-Receptor Antagonist N-(3,5-Dichlorophenyl)-N′-[[4-(1H-imidazol-4-ylmethyl)phenyl]-methyl]-urea (SCH 79687) | Journal of Pharmacology and Experimental Therapeutics](https://jpet.aspetjournals.org/content/jpet/305/3/1037/F1.large.jpg)
Pharmacological Characterization of the Novel Histamine H3-Receptor Antagonist N-(3,5-Dichlorophenyl)-N′-[[4-(1H-imidazol-4-ylmethyl)phenyl]-methyl]-urea (SCH 79687) | Journal of Pharmacology and Experimental Therapeutics

Positively selected modifications in the pore of TbAQP2 allow pentamidine to enter Trypanosoma brucei | bioRxiv

Targeting the Hinge Glycine Flip and the Activation Loop: Novel Approach to Potent p38α Inhibitors | Journal of Medicinal Chemistry
![42206-94-0 | trans-Resveratrol Triacetate | Triacetyl Resveratrol; 3,4',5-Triacetoxy-trans-stilbene; 3,5,4'-Tri-O-acetylresveratrol; 5-[(1E)-2-[4-(Acetyloxy)phenyl]ethenyl]-1,3-benzenediol1,3-Diacetate; Resveratrol 3,5,4'-triacetate; | C20H18O6 | TRC 42206-94-0 | trans-Resveratrol Triacetate | Triacetyl Resveratrol; 3,4',5-Triacetoxy-trans-stilbene; 3,5,4'-Tri-O-acetylresveratrol; 5-[(1E)-2-[4-(Acetyloxy)phenyl]ethenyl]-1,3-benzenediol1,3-Diacetate; Resveratrol 3,5,4'-triacetate; | C20H18O6 | TRC](https://www.trc-canada.com/prod-img/R150055.png)
42206-94-0 | trans-Resveratrol Triacetate | Triacetyl Resveratrol; 3,4',5-Triacetoxy-trans-stilbene; 3,5,4'-Tri-O-acetylresveratrol; 5-[(1E)-2-[4-(Acetyloxy)phenyl]ethenyl]-1,3-benzenediol1,3-Diacetate; Resveratrol 3,5,4'-triacetate; | C20H18O6 | TRC
![PDF] Analgesic effect of GT-0198, a structurally novel glycine transporter 2 inhibitor, in a mouse model of neuropathic pain. | Semantic Scholar PDF] Analgesic effect of GT-0198, a structurally novel glycine transporter 2 inhibitor, in a mouse model of neuropathic pain. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/c6e4767ba53aaf7b60b7691392e038c1fbf1cd7e/2-Figure1-1.png)
PDF] Analgesic effect of GT-0198, a structurally novel glycine transporter 2 inhibitor, in a mouse model of neuropathic pain. | Semantic Scholar