Lab 8 Sodium Carbonate or Sodium Bicarbonate? Objective To determine a compound to be either Na 2 CO 3 or NaHCO ppt download

In the mixture of (NaHCO3 + Na2CO3) volume of HCl required is x mL with phenolphthalein indicator and y mL with methyl orange indicator in the same titration. Hence, volume for complete

Sodium Carbonate + Hydrochloric Acid - Na2CO3 + HCl - Molecular Equations & Net Ionic Equations - YouTube
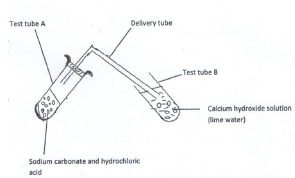
Study the diagram below for the reaction between sodium carbonate and hydrochloric acid and answer the questions that follow
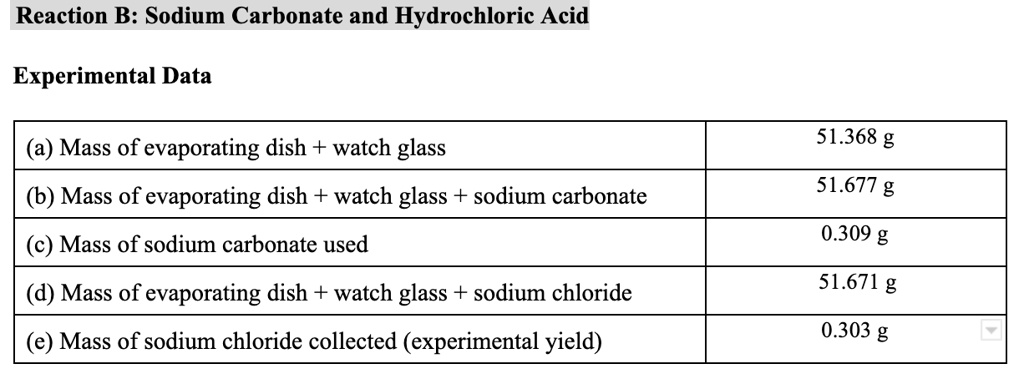
SOLVED: Reaction B: Sodium Carbonate and Hydrochloric Acid Experimental Data Mass of evaporating dish + watch glass 51.368 g Mass of evaporating dish + watch glass + sodium carbonate 51.677 g Mass

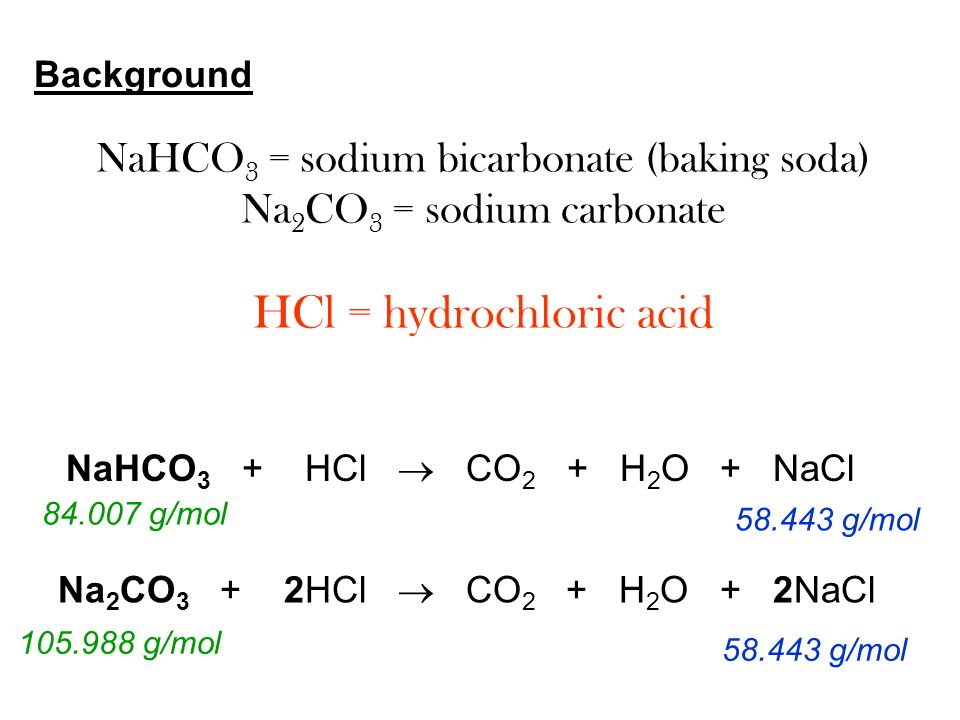
Write the balanced chemical equations for the following reactions:Sodium hydrogen carbonate on reaction with hydrochloric acid gives sodium chloride, water and liberates carbon dioxide.
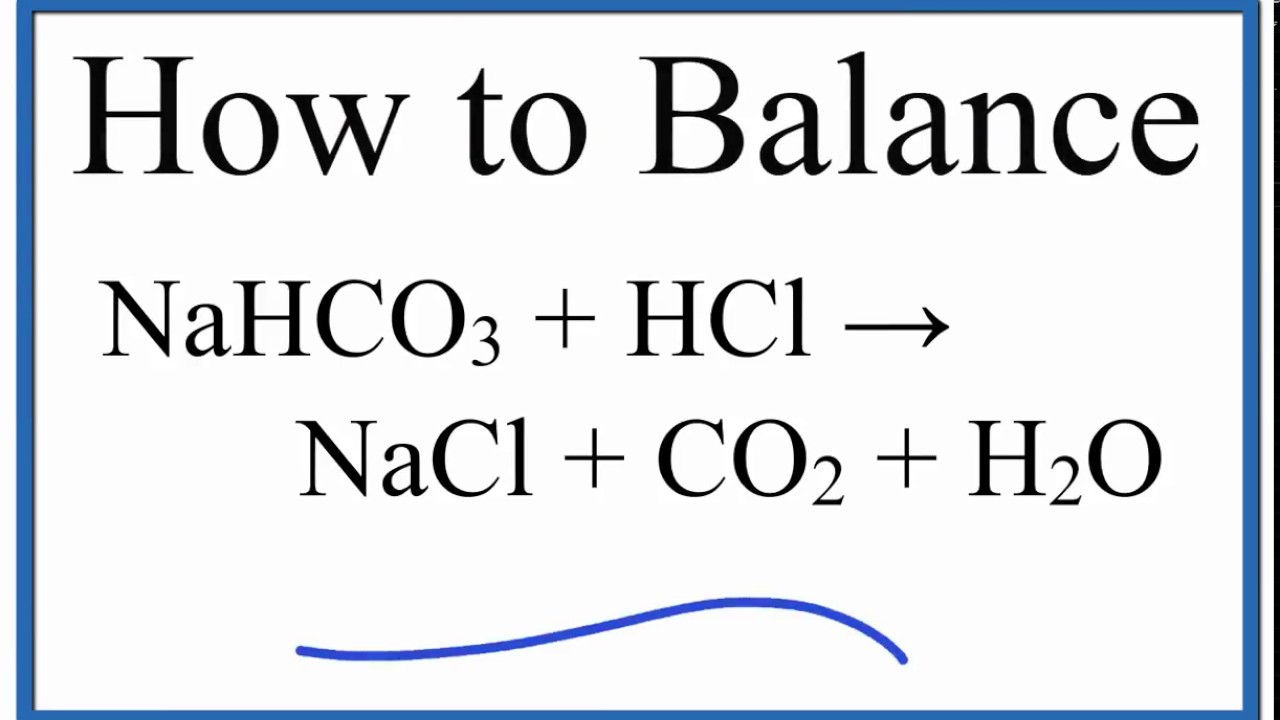
How to Balance NaHCO3 + HCl = NaCl + CO2 + H2O (sodium bicarbonate plus hydrochloric acid) - YouTube

a). What happens when dilute hydrochloric acid is added to sodium carbonate? Write a balanced - YouTube

35. Answer the following questions: (1) State the reaction of sodium carbonate and sodium hydrogen - Brainly.in

Sodium Carbonate + Hydrochloric Acid - Na2CO3 + HCl - Molecular Equations & Net Ionic Equations - YouTube

During the titration of sodium carbonate with H Cl, the dissolved carbonate ion will exist in three different forms; CO_3^{-2}, H CO_3^{-1}, and H_2 CO_3. During which part of the titration (initial,
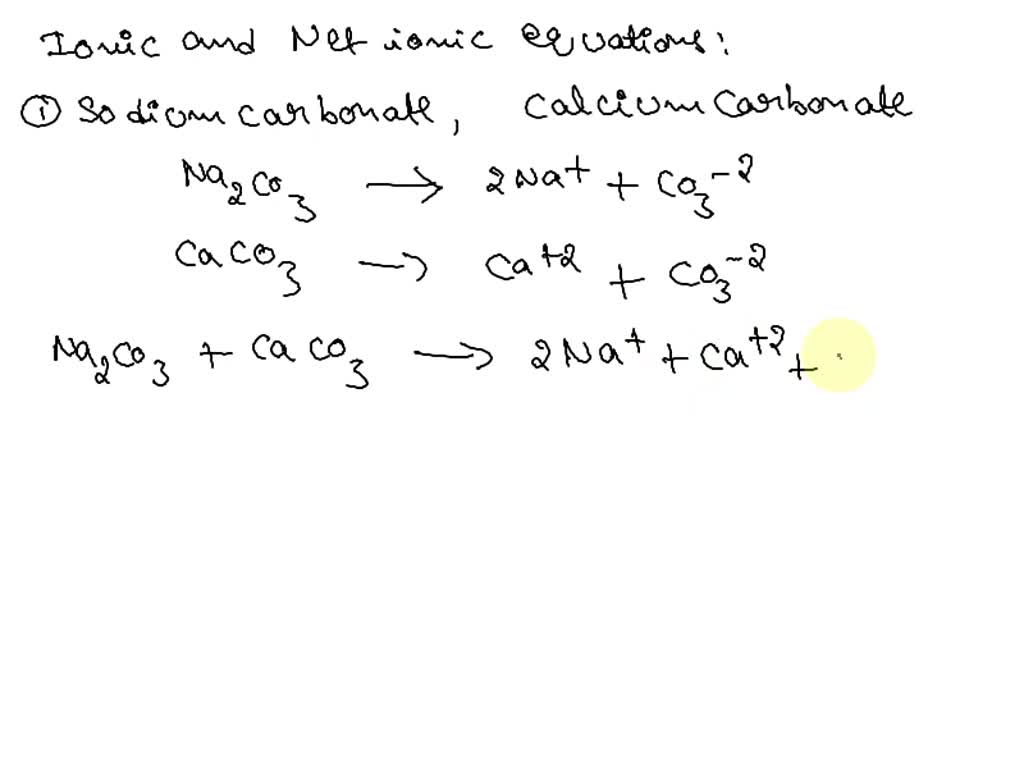
SOLVED: Sodium Carbonate Reactions Sodium Carbonate Reaction with Sodium Hydroxide Full Balanced Chemical Equation Products and the Solubilities Net Ionic Equation Sodium Carbomate Reaction with Zinc Chloride Full Balanced Chemical Equation Products
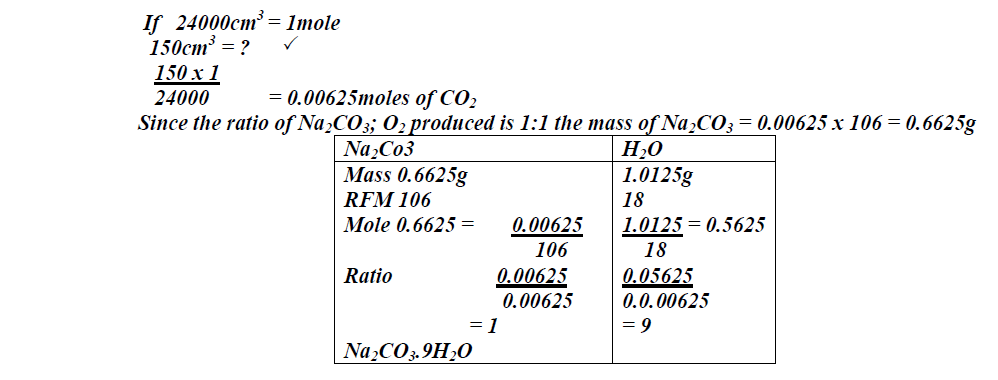
When 1.675g of hydrated sodium carbonate was reacted with excess hydrochloric acid, the volume carbon (IV) oxide gas obtained at room temperature and pressure was 150cm<sup>3</sup>). Calculate...

Sodium Carbonate + Hydrochloric Acid - Balanced Molecular and Net Ionic Equation - Na2CO3 + HCl | Quizalize

![ANSWERED] Sodium carbonate and hydrochloric acid re... - Physical Chemistry ANSWERED] Sodium carbonate and hydrochloric acid re... - Physical Chemistry](https://media.kunduz.com/media/sug-question/raw/51622204-1659180079.3710194.jpeg)