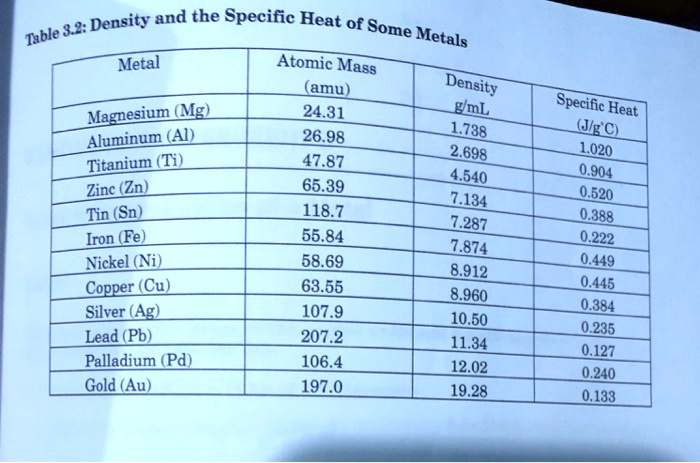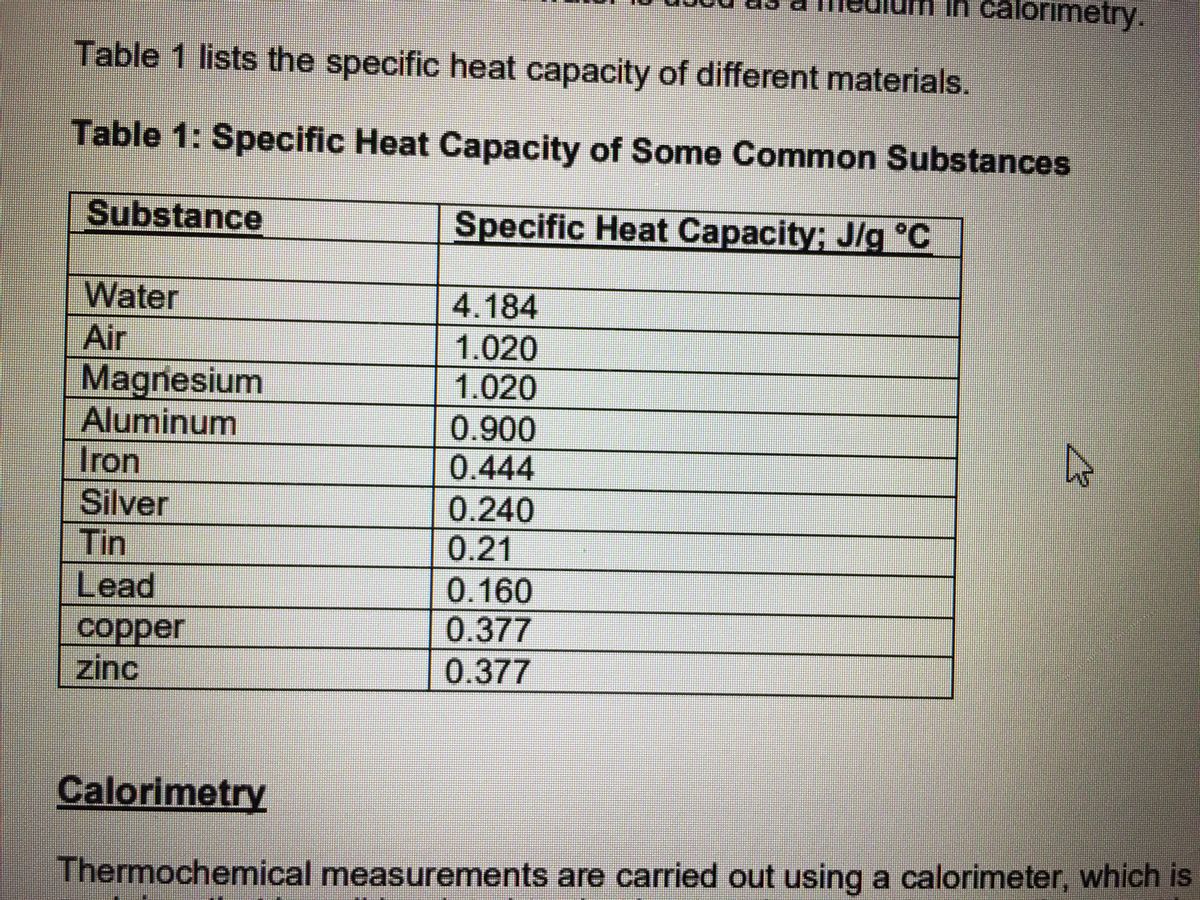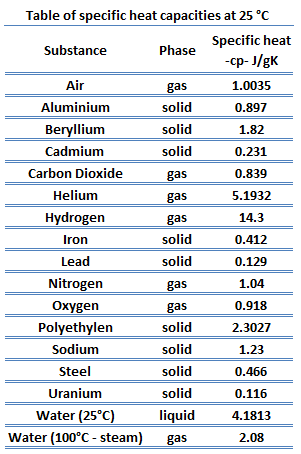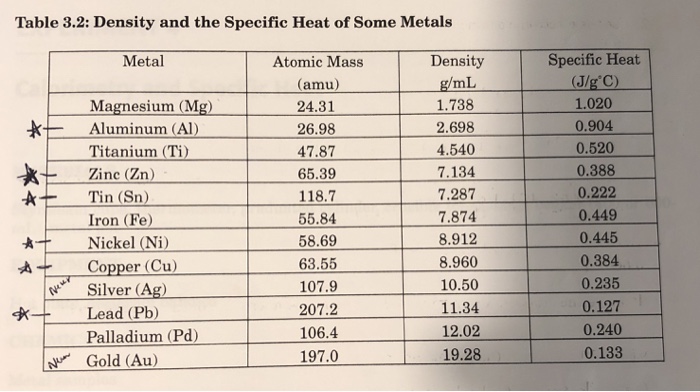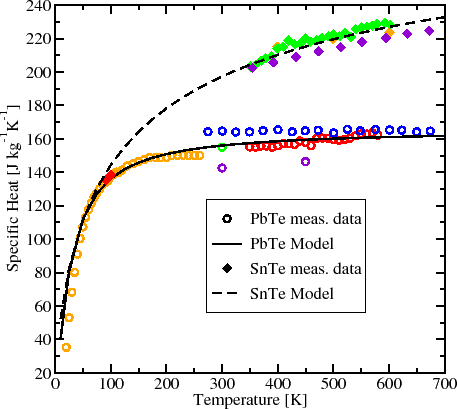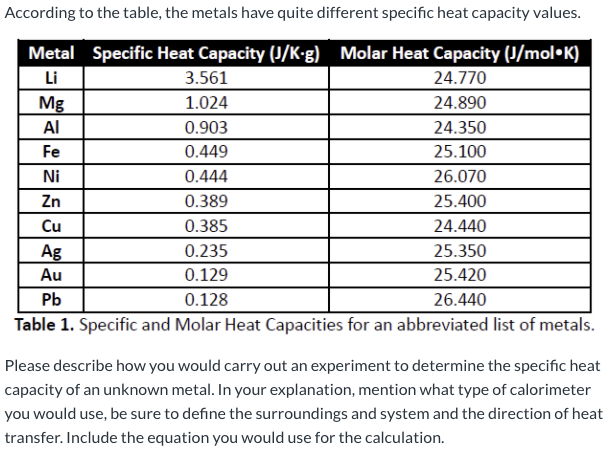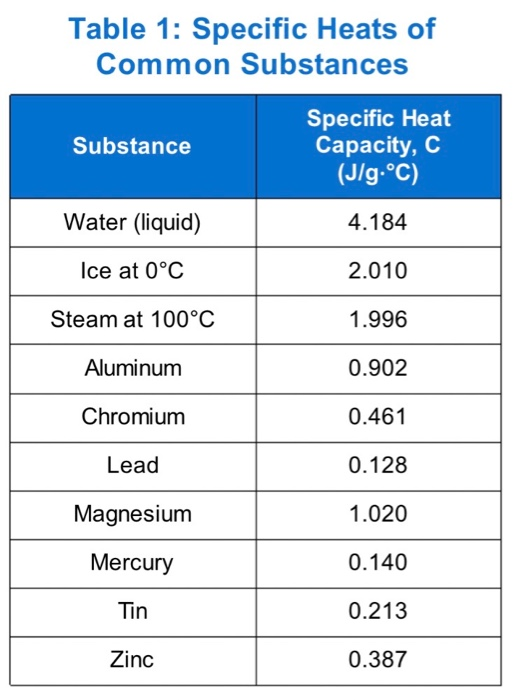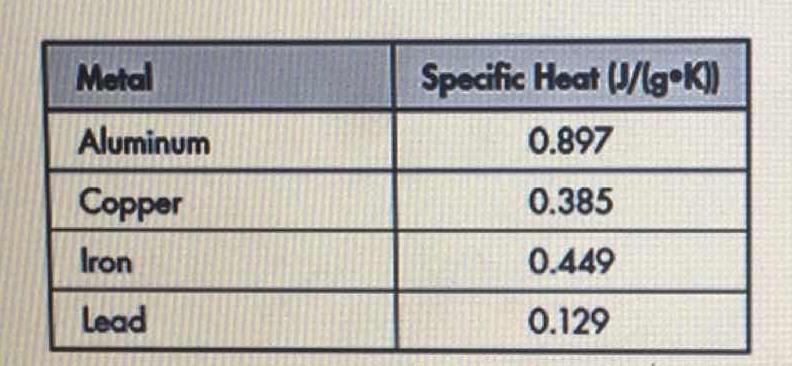
The table below shows the specific heats of several metals. The temperature of a 15-g sample of an unknown metal increases from 20.0 C to 30.0 C when it absorbs 67.5 J

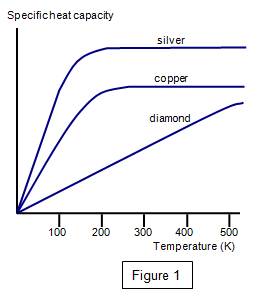
Compare the specific heats of the metals with their atomic weights (i.e., molar masses). Do you see any trends? Make a plot to show this relationship. What does the plot reveal?

SOLVED: Table 1. Specific heat capacities and densities of common metals Metal Specific Heat Capacity 1.g deg Density (gicm ' ) Iron 0.45 7.87 Aluminum 0.91 2.70 Lead 0.13 11.36 Copper 8.96 Zinc 7.13 Tin 0.21 7.28 Gold 19.32

LAB: Specific Heat of a Metal. Prelab question: MetalSpecific Heat (J/g ºC) Aluminum0.91 Iron0.46 Lead0.13 Silver0.23 Tin0.21 Titanium0.54 Zinc0.39 A. - ppt download